More male sex enhancement supplements are recalled because of unlisted ingredients

The U.S. Food and Drug Administration has found another company’s male sexual enhancement supplements could be unsafe for some and has instituted a recall.
On Monday, April 5, the FDA sent an alert that New Jersey’s QMART recalled all lots of its Imperial Gold 2000, PremierZEN Extreme 3000, Burro en Primavera 60000 and Imperial Platinum 2000 products.
What’s the problem?
The FDA’s analysis found that QMART’s supplements were tainted with undeclared sildenafil and/or tadalafil.
These are ingredients known as phosphodiesterase, or PDE-5 inhibitors for short, and are found in FDA-approved products for the treatment of male erectile dysfunction.
The problem is that while those ingredients are approved in products like Viagra and Cialis, those drugs have been tested and approved by the FDA. They also carry warnings for those who might be harmed by the inhibitors.
The FDA isn’t opining on whether the QMART supplements perform as advertised — or don’t — but that the undeclared presence of sildenafil or tadalafil “renders them unapproved drugs for which the safety and efficacy have not been established.”
Consumers with underlying medical issues could experience serious health risks, the FDA said.
“For example, PDE-5 inhibitors may interact with nitrates found in some prescription drugs (such as nitroglycerin) lowering blood pressure to dangerous levels that may be life-threatening. Consumers with diabetes, high blood pressure, or heart disease often take nitrates,” the FDA said.
QMART had not received any reports of adverse events when it issued its notice.
What to look for
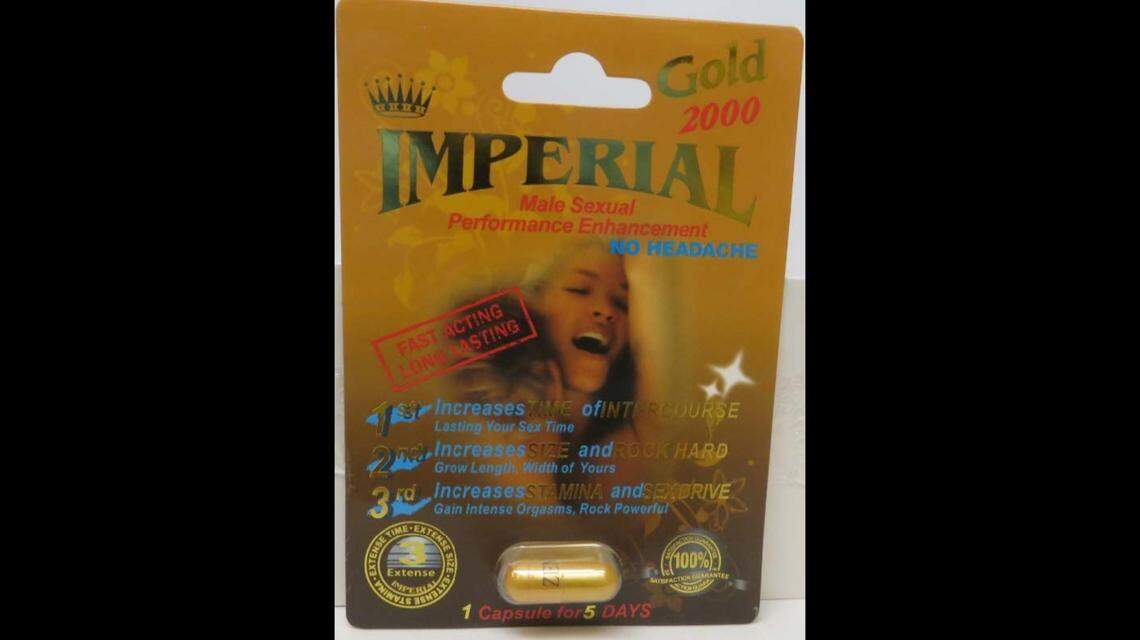

The tainted products are marketed as dietary supplements for male sexual performance enhancement and are packaged in cardboard blisters. All lots of Imperial Gold 2000 and Imperial Platinum 2000, PremierZEN Extreme 3000 and Burro en Primavera 60000 were distributed nationally through internet sales and also fulfilled by Amazon.

If you bought these supplements stop using and discard them, the FDA suggests. Consumers with questions regarding this recall can contact QMART at 551-252-8322 from 9 a.m. to 6 p.m. eastern or email cripsyshopny@gmail.com. If you experienced any problems that may be related to these products contact your doctor or healthcare provider.
Adverse reactions or quality problems experienced with the use of these product can also be reported to the FDA’s MedWatch Adverse Event Reporting program online, by mail or by fax. Call 800-332-1088 if you need to request a reporting form.
Similar sex pill recalls
This is at least the third FDA-inspired recall of male sexual enhancement supplements since December 2019.
On April 1, Nuri Trading, a Korean company that sells male sexual enhancement products online and via Amazon, recalled all lots of Shogun-X 7000, Thumbs Up 7 (Black) 25K, Thumbs Up 7 (White) 11K and 69MODE Blue 69 capsules after the FDA found undeclared tadalafil, sildenafil and/or vardenafi ingredients in the supplements on March 29.
In January 2019, a now dissolved company, Happy Together, issued a recall for the same reason: Its male sexual enhancement drugs, Rhino 5k, had sildenafil and tadalafil.
This story was originally published April 6, 2021 at 9:23 AM with the headline "More male sex enhancement supplements are recalled because of unlisted ingredients."
